Posterior Ankle Impingement - bony
Posterior impingement relates to posterior pain on end-range plantarflexion (PF) due to compression of posterior bony and soft tissue structures (Sofka 2010; Giannini, Buda et al. 2013). It can be caused by overuse, such as ballet dancing or football with repetitive end-range PF, or acute trauma (van Dijk 2006). Although there are many causes for posterior ankle impingement, the symptoms and subsequent treatment are quite similar (Roche, Calder et al. 2013).
Bony Impingement
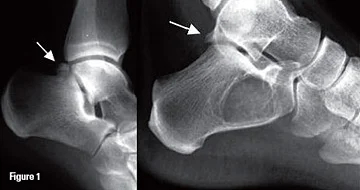
The two most common forms of bony impingement are a Stieda process, an enlarged posterolateral talar process, and an os trigonum, a secondary ossification site of the talus which does not fuse in teenage years remaining as an accessory bone, present in 7 – 14% of adults (Giannini, Buda et al. 2013). An enlarged calcaneal tuberosity can create the same issues (Hess 2011) but is less described in the literature.
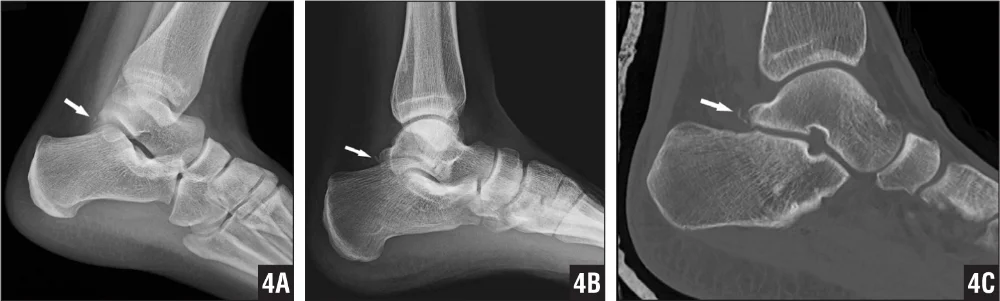
The Stieda process is often asymptomatic but, with repetitive PF loading, can become compacted between the calcaneus and tibia, compressing the posterior soft tissue and causing microtrauma to the bony structures (Niek van Dijk 2006; Hess 2011). With continual compression, chronic inflammation and reduced PF can occur (Hess 2011). Alternatively, an acute impact injury, in the presence of a Stieda process, can cause a fracture which then presents similarly to an os trigonum impingement (Hess 2011).
A patient with a fracture of the Stieda process
Os trigonum impingement occurs when repetitive PF compression irritates local soft tissue launching a chronic inflammatory response (Hess 2011). Scar tissue hypertrophy, joint capsule thickening and excessive inversion or eversion on end-range PF can further compress the os trigonum (Niek van Dijk 2006). Giannini (2013) also describes an avulsion of the os trigonum on forced dorsiflexion, then becoming compressed on PF, placing excessive stress on the posterior talofibular ligament.
The presence of an os trigonum does not automatically indicate pain, there must also be a history of repetitive plantarflexion or forced dorsiflexion, with van Dijk (1995) finding 18 of 38 retired ballet dancers had os trigonums or Stieda processes but experienced no posterior impingement.
Os trigonum (left), Stieda process (right)
Diagnosis
Diagnosis is through subjective questioning and physical examination, with confirmation through investigations and diagnostic injections (Hamilton 2008). The individual generally reports chronic posterior ankle pain on end-range plantarflexion or pushing off activities, for example kicking or wearing high heel shoes (Giannini, Buda et al. 2013), following a history of repetitive overuse or an acute ankle trauma, with tenderness on posterior talar palpation (Niek van Dijk 2006). Giannini (2013) claims individuals compensating for a reduction in plantarflexion may maintain foot inversion on weightbearing, possibly reporting recurrent ankle sprains, calf injury, plantar fasciitis and curling of the toes. A “passive forced plantarflexion test” (Niek van Dijk 2006) is the most sensitive test to implicate posterior impingement, involving quick, repetitive passive end-range plantarflexion oscillations on a patient sitting in 90° knee flexion. Internal and external rotation can be added at end-range plantarflexion, grinding the bony and soft tissue structures, with a negative test clearing posterior impingement (Niek van Dijk 2006). A positive test should then be confirmed through imaging and diagnostic infiltration, with anaesthetic or corticosteroid being injected into the site of bony impingement. If the pain on PF is resolved following injection, a diagnosis of posterior bony ankle impingement can be given (Niek van Dijk 2006).
For treatment, refer to previous blogpost regarding soft tissue ankle impingement.
Differential Diagnosis
Flexor Hallucis Longus Tendinopathy
The flexor hallucis longus (FHL) tendon traverses a fibro-osseous tunnel prior to inserting on the distal hallux, when inflamed within this tunnel posteromedial pain can be produced (Hamilton 2008). Chronic overuse can initiate muscle belly hypertrophy inside the tunnel, causing muscle entrapment and a chronic inflammatory response, ultimately causing FHL stenosing tenosynovitis (Schulhofer and Oloff 2002). The FHL tendon is susceptible to inflammatory symptoms following a singular or repetitive ankle inversion injury, causing medial compression and irritation of the tendon, with poor management increasing the likelihood of developing a tenosynovitis (Hess 2011). In a chronic condition, crepitus, nodule development and calcification of the tendon can occur. Schulhofer and Oloff (2002) state this is commonly seen in ballet dancers who perform continuously in a plantarflexed and toe-extended position, while Hess (2011) describes the presence in athletes requiring a quick push-off action, such as sprinters or hockey players. Once the tendinopathy has initiated, repetitive plantarflexion and inversion further entraps and irritates the tendon (Giannini, Buda et al. 2013).
On assessment, there is pain on palpation of the FHL tendon at the posteromedial ankle, worsened by ankle and hallux dorsiflexion (Schulhofer and Oloff 2002). Michelson and Dunn (2005) propose the “FHL stretch test” as effective in determining the length and irritability of the FHL tendon; the range of hallux dorsiflexion is assessed in both ankle plantarflexion and dorsiflexion. There should be movement at the hallux metatarsophalangeal joint in both positions, if no hallux dorsiflexion occurs when the ankle is dorsiflexed, a tight FHL is present (Michelson and Dunn 2005). MRI is necessary to diagnose tenosynovitis or tendinopathy, as well as excluding bony pathology, other soft tissue structures and joint disruption (Schulhofer and Oloff 2002).
Treatment FHL Tendinopathy
Treatment of FHL tendinopathy, in the literature, consists of rest, immobilisation, deep tissue mobilisation, corticosteroids, non-steroidal anti-inflammatories and physiotherapy, with limited success reported (Schulhofer and Oloff 2002). Hamilton (2008) proposes initial immobilisation to offload the tendon and break the chronic inflammatory cycle, with non-steroidal anti-inflammatories and corticosteroid injections into the tendon sheath if indicated. However recent evidence regarding tendon injuries contradicts this proposed method. Currently the literature does not provide a valid treatment regime specifically for FHL, however adopting the principles from Jill Cook’s tendinopathy research which focuses mostly on the Achilles tendon (Alfredson and Cook 2007), and addressing the inflammatory, strength, endurance and return to function aspects of tendon healing, appears a more evidence-based strategy to resolve this condition.
References
** Giannini, S., Buda, R., Mosca, M., Parma, A., & Di Caprio, F. (2013). Posterior ankle impingement. Foot & ankle international. / American Orthopaedic Foot and Ankle Society [and] Swiss Foot and Ankle Society, 34(3), 459-465.
** Hess, G. W. (2011). Ankle impingement syndromes: a review of etiology and related implications. Foot & ankle specialist, 4(5), 290-297.
Liu, S. H., & Mirzayan, R. (1993). Posteromedial ankle impingement. Arthroscopy : the journal of arthroscopic & related surgery : official publication of the Arthroscopy Association of North America and the International Arthroscopy Association, 9(6), 709-711.
Maitland, G. D. (1991). Peripheral manipulation (3rd ed.). London ; Boston: Butterworth-Heinemann.
Paterson, R. S., & Brown, J. N. (2001). The posteromedial impingement lesion of the ankle. A series of six cases. The American journal of sports medicine, 29(5), 550-557.
** Roche, A. J., Calder, J. D. F., & Lloyd Williams, R. (2013). Posterior ankle impingement in dancers and athletes. Foot and ankle clinics, 18(2), 301-318.
Sofka, C. M. (2010). Posterior ankle impingement: clarification and confirmation of the pathoanatomy. HSS journal : the musculoskeletal journal of Hospital for Special Surgery, 6(1), 99-101.
van Dijk, C. N. (2006). Hindfoot endoscopy. Foot and ankle clinics, 11(2), 391-414, vii.
van Dijk, C. N., Lim, L. S., Poortman, A., Strubbe, E. H., & Marti, R. K. (1995). Degenerative joint disease in female ballet dancers. The American journal of sports medicine, 23(3), 295-300.
** - key references