A mechanism-based approach to clinical reasoning of pain.
Pain is one of the most common complaints of the patients I see as a musculoskeletal physiotherapist working in a private practice setting. More recently in my clinical practice there has been a large amount of complex patients presenting with chronic back pain with or without leg pain, chronic neck and arm pain, and headaches. Each of these having components of nociceptive pain, neuropathic pain and central sensitisation. This has challenged my understanding of each pain mechanism and my ability to explain to patients what their problem is and educate them about pain management.
Inspired to try understand pain a little bit deeper I am going to write a series of blog posts regarding pain. It will start with the mechanisms of pain and clusters of symptoms predictive of each type, delve further into the mechanism of central sensitisation and how to explain this to others, and finally outline what pain education might involve in a 1:1 private practice setting (when we don't have access to pain management clinics).
So this is the first blog and will cover mostly pain mechanisms, the underlying neurophysiology, and what recent research tells us about the relationship between categories and the clinical presentation.
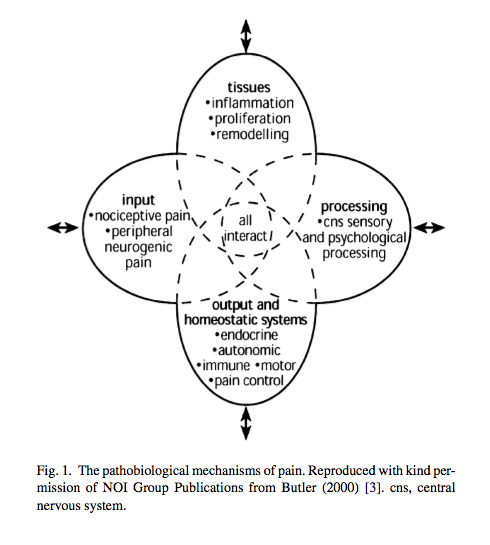
The picture below represents the most commonly accepted categories of pain.
(Smart & Doody, 2006, p. 172)
Smart & Doody (2006) conducted a qualitative study to explore the clinical reasoning process of pain implemented by experienced musculoskeletal physiotherapists. This research from 2006 to 2012 has lead to a greater understanding of our understanding of pain.
Their results indicated that physios use a diverse range of reasoning models which include:
- Biomedical reasoning “included reasoning of pain related to structural/anatomical source, biomechanical principles, aetiology, pathological processes and diagnostic labeling” (K. Smart & Doody, 2007, p. 43).
- Psycholsocial reasoning “included reasoning related to patients’ cognitions, emotions, behaviours, attitudes and coping styles and sociological factors” (Smart & Doody, 2007, p. 44).
- Pain Mechanisms reasoning is “associated with the neurophysiological basis of pain, specifically nociceptive, peripheral neurogenic, central and autonomic/sympathetic mechanisms of pain” (Smart & Doody, 2007, p. 44-45).
- Chronicity
- Irritability/severity
K. Smart, Blake, Staines, and Doody (2010, p. 81) conducted a Delphi study with the aim “to identify those potential clinical criteria upon which clinicians may base such mechanisms-based classifications. They generated an expert-consensus-derived list of clinical criteria associated with a dominance of nociceptive, peripheral neuropathic and central mechanisms of pain. The results were presented in a three-part article which highlighted the following results.
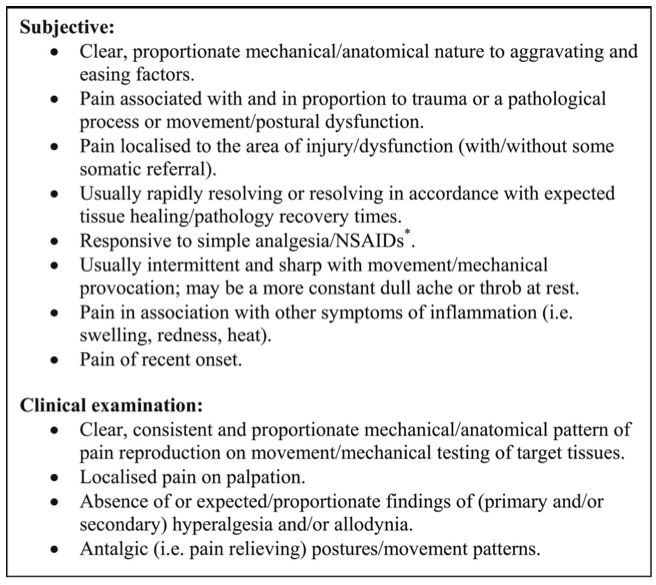
Nociceptive pain.
Nociceptive pain is thought to be a process where the peripheral primary afferent neurones are activated by a noxious stimulus which is either chemical, mechanical or thermal in nature.
Smart et al (2012b) compiled a list of 11 symptoms and four clinical signs (see image below) thought to be predictive of nociceptive pain. Following further analysis from the Delphi study, six symptoms and 1 sign were retained.
- The strongest predictor of nociceptive pain was pain localised to the area of injury/ dysfunction (with/without some somatic referral).
- Clear, proportionate mechanical/anatomical nature to aggravating and easing factors.
- Pain is usually intermittent and sharp with movement/mechanical provocation and may be a more constant dull ache or throb at rest.
- The absence of
- Pain associated with dysesthesia.
- Night pain or sleep disturbances.
- Pain described as burning, shooting, sharp or electric-shock-like.
- Antalgic postures/movement patterns.
The above signs and symptoms had a sensitivity of 90.9%, specificity 91%, positive predictive value of 92.7% and negative predictive value of 88.9%. Overall the positive likelihood ration of 10.10 suggests that patients presenting with this cluster of symptoms are 10 times more likely to have nociceptive pain than peripheral neuropathic or central sensitisation pain.
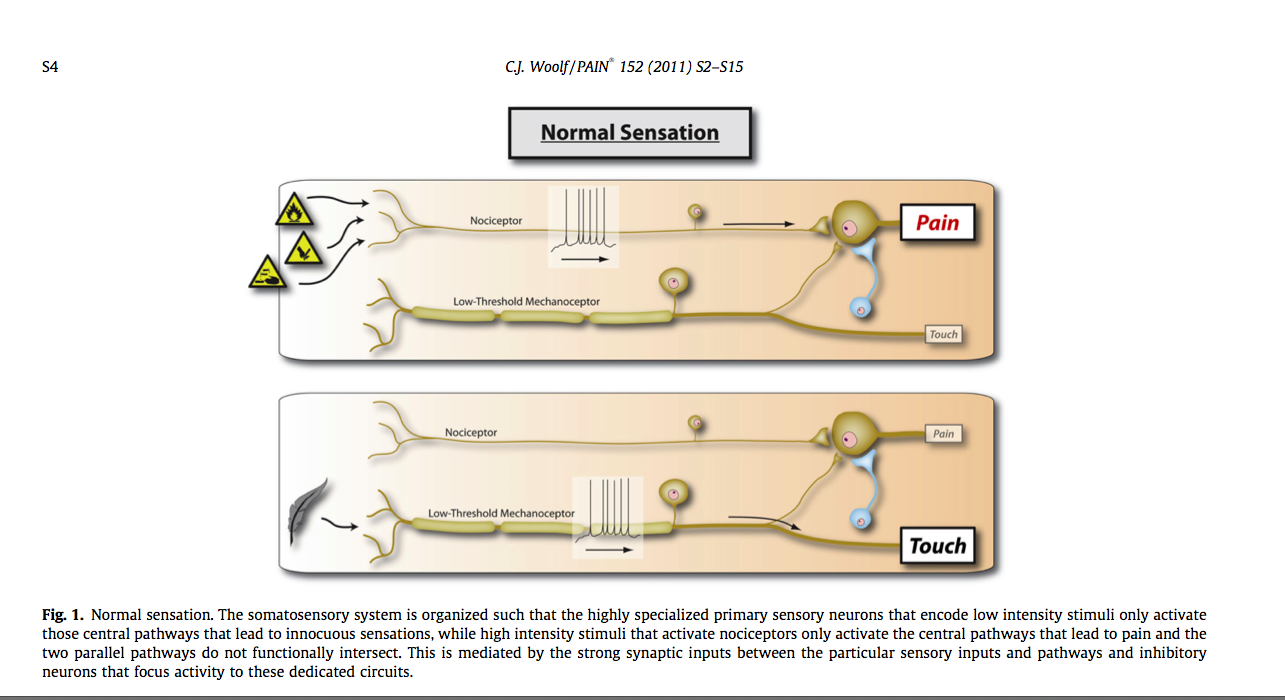
(Woolf, 2011, p. S3)
Peripheral Neurogenic Pain
“Estimates suggest that between 20 and 35% of patients with low back pain (LBP) may have an underlying neuropathic component; and that the costs associated with managing such patients are around 70% higher compared to those with ‘nociceptive’ LBP (Freynhagen and Baron, 2009). Importantly, patients with a dominance of neuropathic pain are known to report more severe pain, greater functional impairments and poorer health related quality of life compared to those with nociceptive/non-neuropathic pain (Smith et al., 2007; Bouhassira et al., 2008; Smart et al., 2012a).” (K. M. Smart, Blake, Staines, Thacker, & Doody, 2012a, p. 345)
So they are harder to manage and improve and the mechanism underlying neuropathic pain is much more complex too....
The underlying process of peripheral neuropathic pain is thought to involve: sensitisation of neural connective tissues, ectopic excitability resulting in spontaneous firing of impulses independent of a peripheral stimulus, cross-excitation resulting in signal amplification, structural changes consisting of axonal sprouting of non-nociceptive fibres resulting in enhancement of nociceptive signalling with non-noxious inputs, and neuro-immune interactions leading to chemical modulation of pain. For further explanation of these mechanisms of peripheral neuropathic pain refer to Smart et al (2012a, p. 346).
K. M. Smart, Blake, et al. (2012a) identified a cluster of two symptoms and one sign predictive of peripheral neuropathic pain:
The strongest predictor of peripheral neuropathic pain was a pain referred in a dermatomal or cutaneous distribution. Patients with this symptom where 24 times more likely to be classified with this pain type.
Patients with a history of nerve injury, pathology or mechanical compromise, were 12 times more likely to be classified with peripheral neuropathic pain, and
Pain/symptom provocation with mechanical/movement tests (e.g. Active/Passive, Neurodynamic) that move/load/compress neural tissue’ meant that patients were 14 times more likely to be classified with peripheral neuropathic pain.
This cluster was found to have high levels of classification accuracy with a sensitivity of 86.3%, specificity of 96%, which “indicates that a patient with the cluster of symptoms and signs outlined by the model was likely to have been classified with peripheral neuropathic pain with an 86.3% level of probability” (K. M. Smart, Blake, et al., 2012a, p. 348).
Neuropathic pain (image from google images).
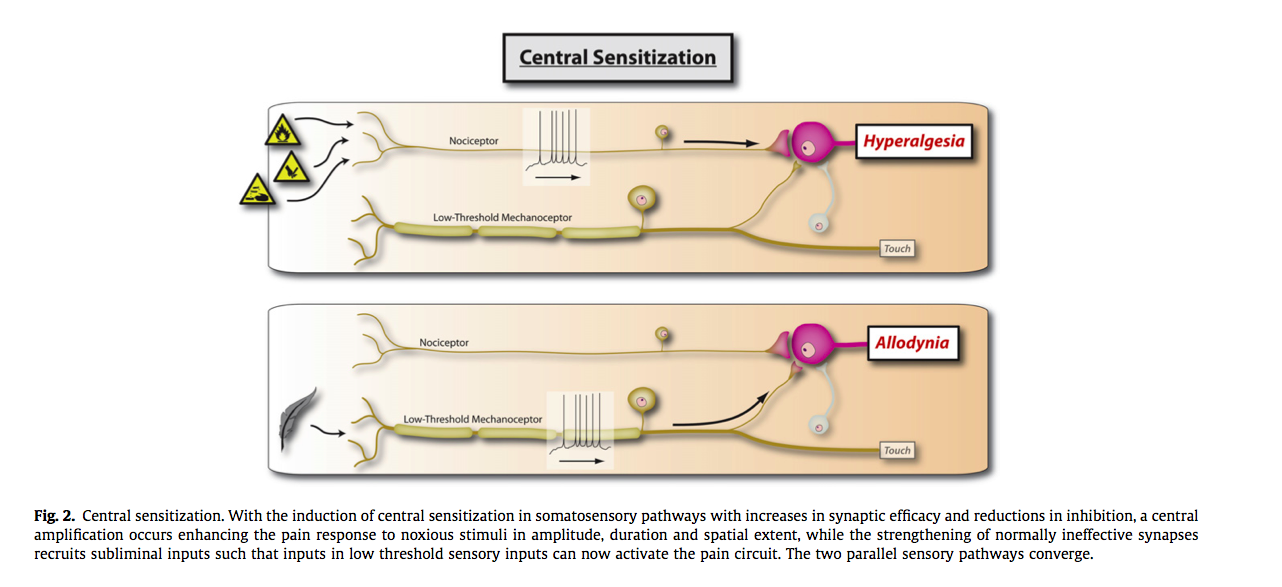
Central sensitisation pain
K. M. Smart, Blake, Staines, and Doody (2012) conducted a study to identify the cluster of subjective symptoms and physical signs that would lead to the ‘mechanism-classification’ of central sensitisation pain.
There were three symptoms and one sign found to be strongly associated with CSP.
- The strongest predictor was disproportionate, non-mechanical, unpredictable pattern of pain provocation in response to multiple/ non-specific aggravating/easing factors.
Pain disproportionate to the nature and extent of the injury or pathology.
Strong association with maladaptive psychosocial factors (e.g. negative emotions, poor self-efficacy, maladaptive beliefs and pain behaviours).
Diffuse/non-anatomic areas of pain/tenderness on palpation. (K. M. Smart, Blake, Staines, & Doody, 2012, p. 342)
This cluster of symptoms was found to have sensitivity of 91.8%, specificity of 97.7%. This can be interpreted as a positive predicative value of 91.8% and negative predictive value of 97.7%. Positive likelihood ration of 40.64 i.e patients with these symptoms are 40 times more likely to have CSP. Overall the predictive accuracy of this cluster of symptoms is very high and therefore can be used clinically to identify patients with CSP.
“Interestingly, a number of symptoms and signs often associated with CSP, such as pain persisting beyond expected tissue healing times and hyperalgesia, did not emerge as predictors of CSP” (Smart, et al., 2012, p. 342).
(Woolf, 2011, p. S4)
Conclusion
The outcome of these delphi studies and the work by Smart and colleagues has provided therapists with clusters of symptoms and clinical signs to assist with the differentiation between, and diagnosis of, different pain mechanisms. This is known to be one of the five models used by therapists during the clinical reasoning process.
Sian
A full list of the origin signs and symptoms are outlined below. Remember that not all of these symptoms have the same predictive accuracy and clinical utility.
Symptoms and clinical features of CENTRAL SENSITISATION PAIN (Smart, Blake, Staines, Thacker, & Doody, 2012, p. 338)
Symptoms and clinical features of NEUROPATHIC PAIN (Smart et al., 2012a, p. 347)
Symptoms and clinical features of NOCICEPTIVE PAIN (Smart et al., 2012b, p 353).
References
Smart, K., Blake, C., Staines, A., & Doody, C. (2010). Clinical indicators of ‘nociceptive’, ‘peripheral neuropathic’ and ‘central’ mechanisms of musculoskeletal pain. A Delphi survey of expert clinicians. Manual therapy, 15(1), 80-87.
Smart, K., & Doody, C. (2006). Mechanisms-based clinical reasoning of pain by experienced musculoskeletal physiotherapists. Physiotherapy, 92(3), 171-178.
Smart, K., & Doody, C. (2007). The clinical reasoning of pain by experienced musculoskeletal physiotherapists. Manual therapy, 12(1), 40-49.
Smart, K. M., Blake, C., Staines, A., & Doody, C. (2012). Mechanisms-based classifications of musculoskeletal pain: part 1 of 3: symptoms and signs of central sensitisation in patients with low back (±leg) pain. Manual therapy, 17(4), 336-344.
Smart, K. M., Blake, C., Staines, A., Thacker, M., & Doody, C. (2012a). Mechanisms-based classifications of musculoskeletal pain: part 2 of 3: symptoms and signs of peripheral neuropathic pain in patients with low back (±leg) pain. Manual therapy, 17(4), 345-351.
Smart, K. M., Blake, C., Staines, A., Thacker, M., & Doody, C. (2012b). Mechanisms-based classifications of musculoskeletal pain: Part 3 of 3: Symptoms and signs of nociceptive pain in patients with low back (±leg) pain. Manual therapy, 17(4), 352-‐357.
Woolf, C. J. (2011). Central sensitization: implications for the diagnosis and treatment of pain. Pain, 152(3), S2-S15.