Pelvic girdle pain - what are we dealing with?
The following three blogs are a review of the most recent clinical practice guidelines for pelvic girdle pain in the antepartum population. Published in 2017, and I believe one of the first of it's kind, particularly in the field of Women's health, this review looks a the body of knowledge prior to 2012. The article by Clinton et al (2017) is a comprehensive evaluation of the literature and provides assessment and treatment recommendations based on the strength of evidence available. What I felt was lacking from this CPG was a guide that would help clinicians improve their framework for assessment an treatment. The aim of this blog series therefore is to delve deeper into the papers behind this review, pull out the finer detail and hopefully provide further clarity to guide clinicians.
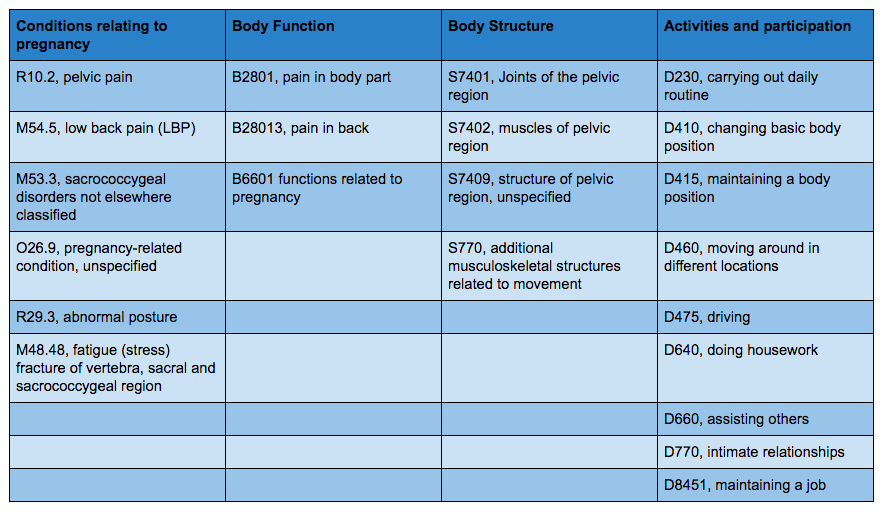
ICD-10 classification codes for PGP
Well this is a first for our blog, because classification codes are unique to Physical Therapy in the United States but also unfamiliar to me as a clinician before I moved here. The reason I have chosen to include this section is because one of the stronger arguments around why we are lacking strong evidence for this conditions is due to variability in nomenclature and terminology. Looking at the list of conditions accepted under this diagnosis below, you can easily see that we have not yet come to a firm conclusion about the language we should be using to identify patients with PGP and manage them. To my understanding, the Australian approach, is to broadly diagnose PGP during pregnancy and often use the terms reduced/excessive force or form closure according to the O’Sullivan classification.
What the table below does represent well is how varied the symptoms are of this condition and the impact it has on the individual within the WHO-ICF framework. Often the name pelvic girdle pain can direct us to focus purely on the pain aspect of the condition and not how it affects the individual's activity levels and participation in daily activities.
Definition
The currently accepted definition for Pelvic Girdle Pain has been continued from the European guidelines published in 2008 and includes the following elements (Vleeming et al., 2008, p 797):
- Pelvic girdle pain generally arises in relation to pregnancy, trauma, arthritis and osteoarthritis.
- Pain is experienced between the posterior iliac crest and the gluteal fold, particularly in the vicinity of the SIJ.
- The pain may radiate in the posterior thigh and can also occur in conjunction with/or separately in the symphysis.
- The endurance capacity for standing, walking, and sitting is diminished.
- The diagnosis of PGP can be reached after exclusion of lumbar causes.
- The pain or functional disturbances in relation to PGP must be reproducible by specific clinical tests.
This same guideline also proposes a functional definition of joint stability (Vleeming, et al., 2009, p. 798):
- ‘‘The effective accommodation of the joints to each specific load demand through an adequately tailored joint compression, as a function of gravity, coordinated muscle and ligament forces, to produce effective joint reaction forces under changing conditions’’.
- Optimal stability is achieved when the balance between performance (the level of stability) and effort is optimized.
- Non-optimal joint stability implicates altered laxity/stiffness values leading to increased joint translations resulting in a new joint position and/or exaggerated/reduced joint compression, with a disturbed performance/effort ratio.
Prevalence
The current CPG states that the "prevalence of PLBP and PGP is estimated to occur in 56% to 72% of the antepartum population, with 20% reporting severe symptoms during 20 to 30 weeks of gestation. In total, 33% to 50% of pregnant females report PGP before 20 weeks of gestation and the prevalence may reach 60% to 70% in late pregnancy.” (Clinton, et al., 2017, p. 107).
This is an incredibly high prevalence!
I thought this number to be so high that I decided to look at two of the key references supporting this claim (Albert et al., 2002; Mens et al., 2012).
“The lack of definition together with a large variety of study designs have led to the fact that the reported incidence of pelvic and low back pain in pregnancy varies between 4% and 76.4%.” (Albert et al., 2002, p.2831). Albert et al (2002) conducted a epidemiological study and described 4 different groups of PGP: pelvic girdle syndrome, symphysiolysis, one-sided sacroiliac syndrome, and double-sided sacroiliac syndrome. They found that many studies did not provide specific detail of the location of pain and how participants were assessed. What Albert et al (2002) concluded was that the incidence of PGP at 33 weeks gestations was 20% and that 6% of pregnant women pelvic girdle syndrome.
Mens et al (2012) reported a much higher number. “In the present study, about 60% of the women reported pain in the lower back and/or pelvis at that moment of examination or during the previous seven days. The severity of experienced pain and disability can be interpreted as mild and moderate in the majority of cases, and severe in about 20%. Women with LBP during pregnancy had more previous pregnancies, a higher BMI and more often had LBP in the past.”
Overall, while the prevalence stated in this CPG appears high, after reviewing some of the backing literature, it appears that the values have been summed together. The higher values are generally derived from studies with a less structured clinical assessment and ones where pregnant women report the presence of PGP based on self assessment and questionnaires. Regardless, PGP is a highly prevalent and varied condition.
Risk Factors
Risk factors are an element all clinicians are curious about and yet we can't concretely make clinical judgements made purely around this area. But who is more at risk according to the current CPG? Those with(Clinton et al., 2017, p. 107):
- A history of multiparity,
- Joint hypermobility,
- Periods of amenorrhea,
- Increased BMI,
- Hip and/or lower extremity dysfunction including the presence of gluteus medius and pelvic floor muscle dysfunction.
- A prior history of trauma to the pelvis or history of lower back pain,
- Pain during previous pregnancies,
- Work dissatisfaction, and
- Lack of belief in improvement.
One of the key articles referenced (Gutke, et al., 2008, p.304) was a Swedish study that explored the role of muscle function and dysfunction in its relation to pregnancy. Essentially, they wanted to find out if it was a case of the chicken or the egg? Do muscles become dysfunctional during pregnancy or are they pre-existing and contribute to the development of LBP and PGP; which is a similar research question to one asked in other sub-populations of LBP?
The overall structure assessment of LBP and PGP is very similar to what I have been taught, except for the assessment of muscle strength. Gutke et al (2008) assessed muscle function of gait speed, hip extension strength with a dynomometer, back flexion strength with a sit up and back extension strength prone on the table lifting their legs. Some of these strength assessments are particularly hard to complete with a pregnant patient. What I use is single leg stance, side lying external rotation strength, side lying active straight leg raise, bridge strength, single or double leg sit to stand and gait.
What they determined from their assessment is that women with PGP +/_ LBP demonstrated lower endurance on back flexion and extension strength, slower walking speeds and weaker hip extension strength. This weakness was seen early in pregnancy suggesting that it may be pre-existing. These findings strengthen the hypothesis that muscle weakness can contribute to reduced force closure and the development of PGP during pregnancy. From these findings other researchers have used exercises to promote force closure by strengthening these muscle groups with good outcomes.
This was just one of the articles I read but the take home message is that many articles included had completely different assessment and screening processes. Therefore it is hard to know the exact weight each of these risk factors has on the condition and is likely to be based on the individual rather than being a hard rule.
When looking at a systematic review from 2009, Vleeming et al state that there are only 2 strong risk factors associated with pregnancy. “Risk factors for developing PGP during pregnancy are most probably: a history of previous low back pain (OR 1.8–2.2) and previous trauma to the pelvis (OR 2.8). (Vleeming, et al., 2009, p.801).
Pathophysiology
“Vleeming et al (1990; 2008) developed the hypothesis of hormonal and biomechanical factors as potential contributors to PGP. Stabilization of the pelvis during load transfer is achieved by the 2 mechanisms of form closure and force closure. Form closure is achieved when the wedge-shaped sacrum fits tightly between the ilia. This process is maximized by the force closure of the muscles, fascia, and ligaments to provide the joint stability. Changes in the ability to manage load transfers due to joint laxity may account for the development of PGP in this population. A change in adequate force and/or form closure of the pelvic girdle was previously postulated to occur by the presence of the hormone relaxin; however, current studies suggest no correlation between relaxin and PGP.” (Clinton, et al., 2017, p.110).
Alicia has previously written about the pathophysiology of PGP and discussed these hormonal changes in further depth. Many physiological changes occur during pregnancy. The most commonly discussed is the change in levels of the sex hormone relaxin. Earlier studies suggest these sex hormones have a role in altering collagen production affecting the ligamentous structures stabilising the pelvic girdle, however recent strong evidence indicates relaxin is not the cause of PGP (Aldabe et al, 2012; Kristiansson et al, 1999). Instead, O’Sullivan and Beales (2007a) propose the existence of other hormones in influencing pain modulation and the inflammatory process, possibly inducing heightened pain reception and prolonging the inflammatory response (Aloisi & Bonifazi, 2006).
Another point to remember is that chronic injury is associated with psychological and cortical changes. O’Sullivan and Beales (2007a) specifically identify the presence of hyper-vigilance, fear avoidance behaviour, altered coping strategies, anxiety, pacing, depression and helplessness in pelvic girdle pain, among others.
Many therapists have identified fear avoidance as a contributor in pelvic girdle pain, affecting the transfer of load through the pelvis. Although not a primary mechanism for the pain, it can cause secondary alterations in muscle activation and motor control, perpetuating the asymmetrical pelvic stability associated with pelvic girdle pain (Beales, O'Sullivan, & Briffa, 2010).
These psychosocial issues have an ability to drastically alter the patient’s outcome, while these factors are not the primary mechanism of pain, they have the capacity to sensitise or amplify pain receptors and disability (O'Sullivan & Beales, 2007a). A patient with poor coping strategies, in the presence of pelvic girdle pain, may experience increased anxiety levels, damaging beliefs, fear avoidance and catastrophising causing a heightened pain response through central sensitisation, further perpetuating the pain cycle (O'Sullivan & Beales, 2007a).
Postural changes
This is a topic everyone loves to talk about. Spoiler Alert! The results are similar to what we know about other conditions too...
- The pain levels cannot be used as a predictor for the width of the pubic symphysis, indicating a relationship between width and pain is present but other pathophysiological processes are also occurring (Bjorklund et al., 2000).
- Damen et al (2001) found a strong correlation between unilateral sacroiliac joint laxity and PGP during and following pregnancy, although this was not bilateral. It appears the laxity is a result of unilateral motor dyskinesis rather than global ligamentous change following pregnancy.
- Hungerford et al (2003) found a delay in muscle activation of multifidus, internal obliquus and gluteus maximus on the stance leg when performing the Stork test, in individuals with sacroiliac joint pain, again proposing an altered stabilisation strategy on pelvic loading.
“The magnitude of postural changes during pregnancy was not indicative of the intensity of PLBP and PGP in the antepartum population.” (Franklin & Conner-Kerr., 1998, p. 137)
While there is ample literature that shows the increase in hormonal levels of relaxin during pregnancy, there is a very poor correlation between range of movement and the development of PGP. It is still poorly understood why PGP begins and why some women can adjust to the hormonal levels without issue, while others cannot (Vleeming, et al., 2009). Just be careful in placing causal blame on ligamentous laxity in the presence of PGP.
Clinical Course
Now that we have sufficiently discussed what our knowledge is about PGP, why it can be limited by strong evidence and what we don't yet know concretely, here are some great facts about the clinical course of PGP that we do know:
- “The most common time period for PGP to occur is between 14 and 30 weeks of gestation.” (Clinton, et al., 2017, p.110).
- “Other factors that also have a high predictive value include a positive posterior PPPT in the first trimester, an increase in the sum scores of compression, distraction, Flexion Abduction External Rotation (FABER) test, and provocative palpation, along with an increase in distress and disability ratings.” (Clinton et al., 2017, p.110).
- The rate of PGP often drops to 7% post-partum (Vleeming, et al, 2009). This review suggest 7-25%, which again is a large variation in range.
- “The incidence of DRA in the antepartum population in the third trimester is 66%, with the occurrence in the postpartum population at 39% after 7 weeks to several years.” (Clinton, et al., 2017, p.110).
- Vollestad & Stuge (2009, p.718) found that scores on the ASLR test and beliefs in improvement (measured on the Oswestry disability index) were predictive regarding the prognosis of PGP post partum. More specifically, “ASLR score <4 predicted 10 points lower ODI and 19 points lower evening pain compared with having ASLR score of at least 4.”
The key points to take from this blog and include in the next step (assessment and diagnosis) are that:
- Pregnancy-related pelvic girdle pain (PGP) is difficult to define as the aetiology and pathoanatomical source of the pain are challenging to localise, with various clinical presentations causing similar pain patterns.
- The is discrepancy in the language used to diagnose lower back pain and SIJ pain during pregnancy.
- Location of pain is very important in assisting diagnosis.
- It is such a highly prevalent conditions, with a complicated etiology and varied clinical course.
- We need to take a thorough history and perform a clinical examination to understand best treatment because generally, the pathogenesis behind PGP is poorly understood.
Sian
References:
Albert, H., Godskesen, M., & Westergaard, J. (2000). Evaluation of clinical tests used in classification procedures in pregnancy-related pelvic joint pain. European Spine Journal, 9(2), 161-166.
Albert, H. B., Godskesen, M., & Westergaard, J. G. (2002). Incidence of four syndromes of pregnancy-related pelvic joint pain. Spine, 27(24), 2831-2834.
Bjorklund, K., Bergstrom, S., Nordstrom, M. L., & Ulmsten, U. (2000). Symphyseal distention in relation to serum relaxin levels and pelvic pain in pregnancy. Acta obstetricia et gynecologica Scandinavica, 79(4), 269-275.
Clinton, S. C., Newell, A., Downey, P. A., & Ferreira, K. (2017). Pelvic Girdle Pain in the Antepartum Population: Physical Therapy Clinical Practice Guidelines Linked to the International Classification of Functioning, Disability, and Health From the Section on Women's Health and the Orthopaedic Section of the American Physical Therapy Association. Journal of Women's Health Physical Therapy, 41(2), 102-125.
Cook, C., Massa, L., Harm-Ernandes, I., Segneri, R., Adcock, J., Kennedy, C., & Figuers, C. (2007). Interrater reliability and diagnostic accuracy of pelvic girdle pain classification. Journal of Manipulative & Physiological Therapeutics, 30(4), 252-258.
Franklin, M. E., & Conner-Kerr, T. (1998). An analysis of posture and back pain in the first and third trimesters of pregnancy. Journal of Orthopaedic & Sports Physical Therapy, 28(3), 133-138.
Gutke, A., Östgaard, H. C., & Öberg, B. (2008). Association between muscle function and low back pain in relation to pregnancy. Journal of rehabilitation medicine, 40(4), 304-311.
Hungerford, B., Gilleard, W., & Hodges, P. (2003). Evidence of altered lumbopelvic muscle recruitment in the presence of sacroiliac joint pain. Spine, 28(14), 1593-1600.
Hungerford, B., Gilleard, W., & Lee, D. (2004). Altered patterns of pelvic bone motion determined in subjects with posterior pelvic pain using skin markers. Clinical biomechanics (Bristol, Avon), 19(5), 456-464.
Mens, J. M., Pool-Goudzwaard, A., & Stam, H. J. (2009). Mobility of the pelvic joints in pregnancy-related lumbopelvic pain: a systematic review. Obstetrical & gynecological survey, 64(3), 200-208.
Mens, J. M., Huis, Y. H., & Pool-Goudzwaard, A. (2012). Severity of signs and symptoms in lumbopelvic pain during pregnancy. Manual therapy, 17(2), 175-179.
Mens, J. M., Vleeming, A., Snijders, C. J., Koes, B. W., & Stam, H. J. (2001). Reliability and validity of the active straight leg raise test in posterior pelvic pain since pregnancy. Spine, 26(10), 1167-1171.
Mens, J. M., Vleeming, A., Snijders, C. J., Koes, B. W., & Stam, H. J. (2002). Validity of the active straight leg raise test for measuring disease severity in patients with posterior pelvic pain after pregnancy. Spine, 27(2), 196-200.
Mens, J. M. A., Snijders, C. J., & Stam, H. J. (2000). Diagonal trunk muscle exercises in peripartum pelvic pain: a randomized clinical trial. Physical therapy, 80(12), 1164-1173.
Nascimento, S. L., Surita, F. G., & Cecatti, J. G. (2012). Physical exercise during pregnancy: a systematic review. Current Opinion in Obstetrics and Gynecology, 24(6), 387-394.
O’Sullivan, P. B., Beales, D. J., Beetham, J. A., Cripps, J., Graf, F., Lin, I. B., ... & Avery, A. (2002). Altered motor control strategies in subjects with sacroiliac joint pain during the active straight-leg-raise test. Spine, 27(1), E1-E8.
O'Sullivan, P., & Beales, D. (2007a). Diagnosis and classification of pelvic girdle pain disorders--Part 1: a mechanism based approach within a biopsychosocial framework. Manual therapy, 12(2), 86-97.
O'Sullivan, P., & Beales, D. (2007b). Diagnosis and classification of pelvic girdle pain disorders, Part 2: illustration of the utility of a classification system via case studies. Manual therapy, 12(2), e1-12.
Vleeming, A., Albert, H. B., Östgaard, H. C., Sturesson, B., & Stuge, B. (2008). European guidelines for the diagnosis and treatment of pelvic girdle pain. European Spine Journal, 17(6), 794-819.
Vleeming, A., Schuenke, M. D., Masi, A. T., Carreiro, J. E., Danneels, L., & Willard, F. H. (2012). The sacroiliac joint: an overview of its anatomy, function and potential clinical implications. Journal of anatomy, 221(6), 537-567.
Vleeming, A., Stoeckart, R., Volkers, A. C., & Snijders, C. J. (1990). Relation between form and function in the sacroiliac joint. Part I: Clinical anatomical aspects. Spine, 15(2), 130-132.
Vøllestad, N. K., & Stuge, B. (2009). Prognostic factors for recovery from postpartum pelvic girdle pain. European spine journal, 18(5), 718-726.